0 of 10 Questions completed Questions: You have already completed the quiz before. Hence you can not start it again.
Quiz is loading… You must sign in or sign up to start the quiz. You must first complete the following:
0 of 10 Questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0) 找出空格里的数字 6 × 5122 ÷ = 2561 找出空格里的数字 6 ÷ × 5122 = 256 找出空格里的数字 7 × 4824 ÷ = 1608 找出空格里的数字 5 ÷ × 6400 = 320 找出空格里的数字 8 × 4528 ÷ = 2264 找出空格里的数字 7 ÷ × 4200 = 210 找出空格里的数字 9 × 7296 ÷ = 1824 找出空格里的数字 8 ÷ × 4000 = 160 找出空格里的数字 5 × 3075 ÷ = 1025 找出空格里的数字 7 ÷ × 8750 = 245
Quiz Summary
Information
Results
Results
0 Essay(s) Pending (Possible Point(s): 0)
Categories
Pos.
Name
Entered on
Points
Result
Table is loading
No data available
1. Question
1 point(s)
2. Question
1 point(s)
3. Question
1 point(s)
4. Question
1 point(s)
5. Question
1 point(s)
6. Question
1 point(s)
7. Question
1 point(s)
8. Question
1 point(s)
9. Question
1 point(s)
10. Question
1 point(s)
Uncategorized
整数的加减混合运算 – 找空格里的数
0 of 10 Questions completed Questions: You have already completed the quiz before. Hence you can not start it again.
Quiz is loading… You must sign in or sign up to start the quiz. You must first complete the following:
0 of 10 Questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0) 找出空格里的数字 + 6421 – 10792 = 17166 找出空格里的数字 38295 – 14923 + = 25591 找出空格里的数字 52983 + 2319 – = 26826 找出空格里的数字 63745 – 35427 + = 33961 找出空格里的数字 + 7435 – 2103 = 21224 找出空格里的数字 48563 – + 10982 = 56800 找出空格里的数字 29587 + – 12345 = 20884 找出空格里的数字 – 2846 + 14322 = 59332 找出空格里的数字 39487 + 5213 – = 27136 找出空格里的数字 50976 – 19876 + = 34724
Quiz Summary
Information
Results
Results
0 Essay(s) Pending (Possible Point(s): 0)
Categories
Pos.
Name
Entered on
Points
Result
Table is loading
No data available
1. Question
1 point(s)
2. Question
1 point(s)
3. Question
1 point(s)
4. Question
1 point(s)
5. Question
1 point(s)
6. Question
1 point(s)
7. Question
1 point(s)
8. Question
1 point(s)
9. Question
1 point(s)
10. Question
1 point(s)
整数的加法 – 找空格里的数
Question: 找出空格里的加数 Instruction: 0 of 10 Questions completed Questions: You have already completed the quiz before. Hence you can not start it again.
Quiz is loading… You must sign in or sign up to start the quiz. You must first complete the following:
0 of 10 Questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0) 找出空格里的加数 27619 + + 34012 = 86769 找出空格里的加数 14567 + + 21852 = 79150 找出空格里的加数 23456 + + 14321 = 67651 找出空格里的加数 17892 + + 28701 = 78200 找出空格里的加数 20234 + + 30760 = 76159 找出空格里的加数 28674 + + 28795 = 74290 找出空格里的加数 13457 + + 26718 = 80037 找出空格里的加数 18452 + + 27619 = 89900 找出空格里的加数 21567 + + 19873 = 76456 找出空格里的加数 19384 + + 14296 = 71100
Answer: 20439 + {31827} + 24314 = 76580
Question
Answer
Steps of Solution
找出空格里的加数
18452 + {43829} + 27619 = 89900
89900 – 18452 – 27619 = 43829
找出空格里的加数
21567 + {35016} + 19873 = 76456
76456 – 21567 – 19873 = 35016
找出空格里的加数
19384 + {37420} + 14296 = 71100
71100 – 19384 – 14296 = 37420
找出空格里的加数
27619 + {25138} + 34012 = 86769
86769 – 27619 – 34012 = 25138
找出空格里的加数
14567 + {42731} + 21852 = 79150
79150 – 14567 – 21852 = 42731
找出空格里的加数
23456 + {29874} + 14321 = 67651
67651 – 23456 – 14321 = 29874
找出空格里的加数
17892 + {31607} + 28701 = 78200
78200 – 17892 – 28701 = 31607
找出空格里的加数
20234 + {25165} + 30760 = 76159
76159 – 20234 – 30760 = 25165
找出空格里的加数
28674 + {16821} + 28795 = 74290
74290 – 28674 – 28795 = 16821
找出空格里的加数
13457 + {39862} + 26718 = 80037
80037 – 13457 – 26718 = 39862
Quiz Summary
Information
Results
Results
0 Essay(s) Pending (Possible Point(s): 0)
Categories
Pos.
Name
Entered on
Points
Result
Table is loading
No data available
1. Question
1 point(s)
2. Question
1 point(s)
3. Question
1 point(s)
4. Question
1 point(s)
5. Question
1 point(s)
6. Question
1 point(s)
7. Question
1 point(s)
8. Question
1 point(s)
9. Question
1 point(s)
10. Question
1 point(s)
整数的加减混合运算 – 找空格里的数 – 1
0 of 10 Questions completed Questions: You have already completed the quiz before. Hence you can not start it again.
Quiz is loading… You must sign in or sign up to start the quiz. You must first complete the following:
0 of 10 Questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0) 找出空格里的数字 + 6421 – 10792 = 17166 找出空格里的数字 38295 – 14923 + = 25591 找出空格里的数字 52983 + 2319 – = 26826 找出空格里的数字 63745 – 35427 + = 33961 找出空格里的数字 + 7435 – 2103 = 21224 找出空格里的数字 48563 – + 10982 = 56800 找出空格里的数字 29587 + – 12345 = 20884 找出空格里的数字 – 2846 + 14322 = 59332 找出空格里的数字 39487 + 5213 – = 27136 找出空格里的数字 50976 – 19876 + = 34724
Quiz Summary
Information
Results
Results
0 Essay(s) Pending (Possible Point(s): 0)
Categories
Pos.
Name
Entered on
Points
Result
Table is loading
No data available
1. Question
1 point(s)
2. Question
1 point(s)
3. Question
1 point(s)
4. Question
1 point(s)
5. Question
1 point(s)
6. Question
1 point(s)
7. Question
1 point(s)
8. Question
1 point(s)
9. Question
1 point(s)
10. Question
1 point(s)
Oxidation and Reduction in Term of Change of Oxidation Number
0 of 10 Questions completed Questions: You have already completed the quiz before. Hence you can not start it again.
Quiz is loading… You must sign in or sign up to start the quiz. You must first complete the following:
0 of 10 Questions answered correctly
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0) Find the oxidation number of sulphur in Na₂S₂O3. What is the oxidation number of sulphur in K₂SO₃? What is the oxidation number of vanadium in V₂O₅? What is the oxidation state of nitrogen in a nitric acid molecule? What is the oxidation state of phosphorus in a phosphoric acid molecule? What is the oxidation number of hydrogen in magnesium hydride, \(\mathrm{MgH}_2\) ? Which of the underlined element in the following compounds has the highest oxidation number? Which of the following is true about the oxidation number of the elements in NH₄ClO₃. What is the oxidation number of \(\mathrm{X}\) in \(\mathrm{XO}_4^{-}\)? What is the oxidation number of the chromium element in potassium dichromate(VI), \(\mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7\) ?
Quiz Summary
Information
Results
Results
0 Essay(s) Pending (Possible Point(s): 0)
Average score
Your score
Categories
Pos.
Name
Entered on
Points
Result
Table is loading
No data available
1. Question
1 point(s)
2. Question
1 point(s)
3. Question
1 point(s)
4. Question
1 point(s)
5. Question
1 point(s)
6. Question
1 point(s)
7. Question
1 point(s)
8. Question
1 point(s)
9. Question
1 point(s)
10. Question
1 point(s)
0 of 7 Questions completed Questions: You have already completed the quiz before. Hence you can not start it again.
Quiz is loading… You must sign in or sign up to start the quiz. You must first complete the following:
0 of 7 Questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0) The following ionic equation represents the reaction between acidified potassium dichromate(VI) solution and iron(II) sulphate solution. The following equation represents the oxidation reaction between ethanol and acidified potassium dichromate(VI). What is the change in the oxidation number of the element chromium in this reaction? Diagram 6 shows the chemical formula of a compound. \begin{align} The following chemical equation represents the extraction of silicon from quartz using coke. The following ionic equation shows the reaction between zinc and acid. Which of the following IUPAC names are correct for the respective formulae of substances? The equation below shows the decomposition of potassium chlorate. 2KClO₃ → 2KCl + 3O₂ Which of the following shows the change of oxidation number of chlorine in the reaction?
Quiz Summary
Information
Results
Results
0 Essay(s) Pending (Possible Point(s): 0)
Categories
Pos.
Name
Entered on
Points
Result
Table is loading
No data available
1. Question
1 point(s)
\[
\mathrm{Cr}_2 \mathrm{O}_7^{2-}+14 \mathrm{H}^{+}+6 \mathrm{Fe}^{2+} \rightarrow 6 \mathrm{Fe}^{3+}+2 \mathrm{Cr}^{3+}+7 \mathrm{H}_2 \mathrm{O}
\]
What is the change of oxidation number of chromium in the reaction?
2. Question
1 point(s)
\[
\begin{aligned}
& 3 \mathrm{C}_2 \mathrm{H}_5 \mathrm{OH}+2 \mathrm{~K}_2 \mathrm{Cr}_2 \mathrm{O}_7+8 \mathrm{H}_2 \mathrm{SO}_4 \rightarrow \\
& \quad 3 \mathrm{CH}_3 \mathrm{COOH}+2 \mathrm{Cr}_2\left(\mathrm{SO}_4\right)_3+2 \mathrm{~K}_2 \mathrm{SO}_4+11 \mathrm{H}_2 \mathrm{O}
\end{aligned}
\]
3. Question
1 point(s)
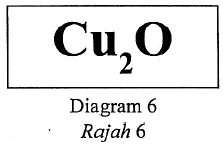
the IUPAC name and oxidation number of copper element in the compound?
\begin{array}{|c|l|c|}
\hline & \text { Name of compound } & \begin{array}{l}
\text { Oxidation number of } \\
\text { copper element }
\end{array} \\
\hline \text { A } & \text { Copper(I) oxide } & +1 \\
\hline \text { B } & \text { Copper(I) oxide } & +2 \\
\hline \text { C } & \text { Copper(II) oxide } & +1 \\
\hline \text { D } & \text { Copper(II) oxide } & +2 \\
\hline
\end{array}
\end{align}
4. Question
1 point(s)
\[
\mathrm{SiO}_2+\mathrm{C} \longrightarrow \mathrm{Si}+\mathrm{CO}_2
\]
What is the change in oxidation number of silicon?
5. Question
1 point(s)
\[
\mathrm{Zn}(\mathrm{s})+2 \mathrm{H}^{+}(\mathrm{aq}) \rightarrow \mathrm{Zn}^{2+}(\mathrm{aq})+\mathrm{H}_2(\mathrm{~g})
\]
What is the change in oxidation number of hydrogen?
6. Question
1 point(s)
7. Question
1 point(s)
0 of 9 Questions completed Questions: You have already completed the quiz before. Hence you can not start it again.
Quiz is loading… You must sign in or sign up to start the quiz. You must first complete the following:
0 of 9 Questions answered correctly
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0) The equation represents the redox reaction between ammonia and copper(II) oxide. 2NH₃ + 3CuO → N₂ + 3Cu + 3H₂O Which of the following is true about the reaction? The oxidation number of calcium increases when Which of the underlined substances in the following equations undergo oxidation? Which of the following statements about a chemical reaction are true? A substance undergoes oxidation if Which of the following indicates that a substance undergoes reduction? Which of the following is NOT true about oxidation and reduction? Which of the underlined substances in the following equations undergo oxidation? The half equation represents the reduction reaction of acidified potassium dichromate(VI) solution. \begin{align}
Quiz Summary
Information
Results
Results
0 Essay(s) Pending (Possible Point(s): 0)
Average score
Your score
Categories
Pos.
Name
Entered on
Points
Result
Table is loading
No data available
1. Question
1 point(s)
2. Question
1 point(s)
3. Question
1 point(s)
4. Question
1 point(s)
5. Question
1 point(s)
6. Question
1 point(s)
7. Question
1 point(s)
8. Question
1 point(s)
9. Question
1 point(s)
\[
\mathrm{Cr}_2 \mathrm{O}_7^{2-}+\mathrm{xH}^{+}+\mathrm{ye}^{-} \longrightarrow \mathrm{zCr}^{3+}+7 \mathrm{H}_2 \mathrm{O}
\]
What are the value of \(x, y\) and \(z\) ?
\begin{array}{|c|c|c|c|}
\hline & x & y & z \\
\hline \text { A } & 14 & 6 & 2 \\
\hline \text { B } & 14 & 5 & 1 \\
\hline \text { C } & 7 & 2 & 1 \\
\hline \text { D } & 7 & 1 & 2 \\
\hline
\end{array}
\end{align}
Redox Reaction
0 of 7 Questions completed Questions: You have already completed the quiz before. Hence you can not start it again.
Quiz is loading… You must sign in or sign up to start the quiz. You must first complete the following:
0 of 7 Questions answered correctly
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0) Reducing agents are ____1____ in a reaction. Reducing agents cause the other substance in the reaction to ____2____ oxygen. Which words complete gaps 1 and 2? Magnesium reacts with copper oxide. The equation for this reaction is shown. Mg+CuO→MgO+Cu Which substance is acting as an oxidising agent in this reaction? Iron is extracted from its oxide using carbon monoxide. The equation is shown. Fe₂O₃+3CO→2Fe+3CO₂ Which row identifies the reducing agent and explains how it acts as a reducing agent? Iron can be obtained from iron(III) oxide by heating with aluminium powder. The equation is shown. 2Al+Fe₂O₃→Al₂O₃+2Fe What is the oxidising agent? The equation for the reaction of zinc oxide with carbon monoxide is shown. ZnO+CO→Zn+CO₂ Which statement explains the role of carbon monoxide in this reaction? Iron oxide reacts with aluminium. Fe₂O₃+2Al→2Fe+Al₂O₃ Which row identifies the oxidising agent and reducing agent in the reaction? 2 NH₃ + 3CuO → N₂ + 3Cu + 3H₂O The equation above represents the reaction between ammonia and copper(II) oxide. Which of the following is true about the reaction?
Quiz Summary
Information
Results
Results
0 Essay(s) Pending (Possible Point(s): 0)
Average score
Your score
Categories
Pos.
Name
Entered on
Points
Result
Table is loading
No data available
1. Question
1 point(s)
2. Question
1 point(s)
3. Question
1 point(s)
Reducing agent
How it reacts
A
Fe₂O₃
receives oxygen
B
Fe₂O₃
donates oxygen
C
CO
receives oxygen
D
CO
donates oxygen
4. Question
1 point(s)
5. Question
1 point(s)
6. Question
1 point(s)
7. Question
1 point(s)
0 of 9 Questions completed Questions: You have already completed the quiz before. Hence you can not start it again.
Quiz is loading… You must sign in or sign up to start the quiz. You must first complete the following:
0 of 9 Questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0) Which of the following equation shows that a chlorine molecule receives 2 electrons to form 2 chloride ions? Which of the following equation shows that a copper(II) ion receive 2 electrons to form Which of the following equation shows that a zinc atom donates 2 electrons to form a zinc ion? The following equation represents the oxidation of magnesium atom. Mg → Mg²⁺ + 2e- What is meant by oxidation based on the equation? Which of the following is a reduction reaction? Which half equations show that reactant acts as a reducing agent? I. \(\mathrm{Mg} \longrightarrow \mathrm{Mg}^{2+}+2 \mathrm{e}^{-}\) The reaction between iron(II) sulphate solution and bromine can be represented by the Fe²⁺ + Cl₂ → Fe³⁺ + 2Cl⁻ Which of the following is true about the reaction? Which of the followings are reducing process? Which set of definition of oxidation in terms of oxygen, hydrogen and electron is correct? \[
Quiz Summary
Information
Results
Results
0 Essay(s) Pending (Possible Point(s): 0)
Categories
Pos.
Name
Entered on
Points
Result
Table is loading
No data available
1. Question
1 point(s)
2. Question
1 point(s)
copper atom?
3. Question
1 point(s)
4. Question
1 point(s)
5. Question
1 point(s)
6. Question
1 point(s)
II. \(\mathrm{Cu}^{2+}+2 \mathrm{e}^{-} \longrightarrow \mathrm{Cu}\)
III. \(2 \mathrm{Cl}^{-} \longrightarrow \mathrm{Cl}_2+2 \mathrm{e}^{-}\)
IV. \(2 \mathrm{H}^{+}+2 \mathrm{e}^{-} \longrightarrow \mathrm{H}_2\)
7. Question
1 point(s)
following equation.
8. Question
1 point(s)
9. Question
1 point(s)
\begin{array}{|l|l|l|l|}
\hline & \text { Oxygen } & \text { Hydrogen } & \text { Electrons } \\
\hline \text { A } & \text { Gain } & \text { Loss } & \text { Loss } \\
\hline \text { B } & \text { Gain } & \text { Loss } & \text { Gain } \\
\hline \text { C } & \text { Loss } & \text { Gain } & \text { Gain } \\
\hline \text { D } & \text { Loss } & \text { Gain } & \text { Loss } \\
\hline
\end{array}
\]
0 of 10 Questions completed Questions: You have already completed the quiz before. Hence you can not start it again.
Quiz is loading… You must sign in or sign up to start the quiz. You must first complete the following:
0 of 10 Questions answered correctly
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0) Find the oxidation number of sulphur in Na₂S₂O3. What is the oxidation number of sulphur in K₂SO₃? What is the oxidation number of vanadium in V₂O₅? What is the oxidation state of nitrogen in a nitric acid molecule? What is the oxidation state of phosphorus in a phosphoric acid molecule? What is the oxidation number of hydrogen in magnesium hydride, \(\mathrm{MgH}_2\) ? Which of the underlined element in the following compounds has the highest oxidation number? Which of the following is true about the oxidation number of the elements in NH₄ClO₃. What is the oxidation number of \(\mathrm{X}\) in \(\mathrm{XO}_4^{-}\)? What is the oxidation number of the chromium element in potassium dichromate(VI), \(\mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7\) ?
Quiz Summary
Information
Results
Results
0 Essay(s) Pending (Possible Point(s): 0)
Average score
Your score
Categories
Pos.
Name
Entered on
Points
Result
Table is loading
No data available
1. Question
1 point(s)
2. Question
1 point(s)
3. Question
1 point(s)
4. Question
1 point(s)
5. Question
1 point(s)
6. Question
1 point(s)
7. Question
1 point(s)
8. Question
1 point(s)
9. Question
1 point(s)
10. Question
1 point(s)
Composite Material
0 of 7 Questions completed Questions: You have already completed the quiz before. Hence you can not start it again.
Quiz is loading… You must sign in or sign up to start the quiz. You must first complete the following:
0 of 7 Questions answered correctly
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0) CHEM0906010 CHEM0906020 CHEM0906030 CHEM0906040 CHEM0906050 CHEM0906060 Which of the following is not correct about the composite material and its property?
Quiz Summary
Information
Results
Results
0 Essay(s) Pending (Possible Point(s): 0)
Average score
Your score
Categories
Pos.
Name
Entered on
Points
Result
Table is loading
No data available
1. Question
1 point(s)
Which of the following statements are true about composite materials?
2. Question
1 point(s)
Which of the following are composite material?
3. Question
1 point(s)
Which of the followings can be made of composite material?
4. Question
1 point(s)
Photochromic glass is used in making
5. Question
1 point(s)
Fibreglass is used to make racquets because it is
6. Question
1 point(s)
Which of the following material is used in the infrastructures of telecommunication?
7. Question
1 point(s)
Glass and Ceramics
0 of 9 Questions completed Questions: You have already completed the quiz before. Hence you can not start it again.
Quiz is loading… You must sign in or sign up to start the quiz. You must first complete the following:
0 of 9 Questions answered correctly
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0) CHEM0905010 CHEM0905020 CHEM0905030 CHEM0905040 CHEM0905050 CHEM0905060 Low coefficient of thermal expansion. What is substance Y? CHEM0905070 CHEM0905080 The image above shows some electric cables supported by some insulator. The insulator is most probably made up of CHEM0905100
Quiz Summary
Information
Results
Results
0 Essay(s) Pending (Possible Point(s): 0)
Average score
Your score
Categories
Pos.
Name
Entered on
Points
Result
Table is loading
No data available
1. Question
1 point(s)
Which of the following glass is normally used to make windows, mirrors and bottles?
2. Question
1 point(s)
Which of the following are the characteristics of ceramic?
3. Question
1 point(s)
Which of the following are the common properties of glass and ceramic?
4. Question
1 point(s)
What type of glass is formed when calcium carbonate is heated with silicon dioxide?
5. Question
1 point(s)
Ceramic is used as the wall of nuclear reactors because
6. Question
1 point(s)
Substance Y has the following properties.
Very high resistance to chemical substances.
7. Question
1 point(s)
A substance X is added to soda lime glass in the manufacturing process so that the glass is more resistance to heat. What is X?
8. Question
1 point(s)

9. Question
1 point(s)
Which of the following glass is resistance to heat, transparent to infrared but not transparent to UV light?
Latihan
0 of 11 Questions completed Questions: You have already completed the quiz before. Hence you can not start it again.
Quiz is loading… You must sign in or sign up to start the quiz. You must first complete the following:
0 of 11 Questions answered correctly
Your time:
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0) CHEM0404010 Manakah halogen berikut wujud sebagai cecair merah jambu pada suhu bilik? CHEM0404020 Antara berikut yang manakah paling reaktif secara kimia dalam kumpulan halogen? CHEM0404030 Manakah antara pernyataan berikut BENAR tentang unsur Kumpulan 17? CHEM0404040 Manakah antara pernyataan berikut adalah BENAR tentang unsur Kumpulan 17? CHEM0404050 Antara berikut, yang manakah betul tentang ciri-ciri unsur dalam Kumpulan 17 dalam jadual berkala apabila menuruni kumpulan? CHEM0404060 Klorin dan bromin adalah unsur dalam Kumpulan 17 dalam jadual berkala. Takat didih klorin lebih rendah daripada bromin. Pernyataan yang manakah di bawah ini menerangkan fenomena ini? CHEM0404070 Unsur X terletak dalam kumpulan yang sama dengan bromin dalam jadual berkala unsur. Manakah antara berikut yang benar tentang unsur X? CHEM0404080 Unsur P larut dalam air untuk menghasilkan larutan asid. Larutan ini boleh mengubah kertas litmus menjadi tidak berwarna. Apakah unsur yang paling mungkin menjadi P? CHEM0404090 Apakah hasil yang terbentuk apabila bromin bertindak balas dengan natrium hidroksida? CHEM0404100 Apakah hasil yang terbentuk apabila gas klorin larut dalam air? CHEM0404110 Apakah kegunaan-kegunaan klorin berikut?
Quiz Summary
Information
Results
Results
0 Essay(s) Pending (Possible Point(s): 0)
Categories
Pos.
Name
Entered on
Points
Result
Table is loading
No data available
1. Question
1 point(s)
2. Question
1 point(s)
3. Question
1 point(s)
4. Question
1 point(s)
5. Question
1 point(s)
6. Question
1 point(s)
7. Question
1 point(s)
8. Question
1 point(s)
9. Question
1 point(s)
10. Question
1 point(s)
11. Question
1 point(s)
Practice
0 of 11 Questions completed Questions: You have already completed the quiz before. Hence you can not start it again.
Quiz is loading… You must sign in or sign up to start the quiz. You must first complete the following:
0 of 11 Questions answered correctly
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0) CHEM0404010 CHEM0404020 CHEM0404030 CHEM0404040 CHEM0404050 CHEM0404060 CHEM0404070 CHEM0404080 CHEM0404090 CHEM0404100 CHEM0404110
Quiz Summary
Information
Results
Results
0 Essay(s) Pending (Possible Point(s): 0)
Average score
Your score
Categories
Pos.
Name
Entered on
Points
Result
Table is loading
No data available
1. Question
1 point(s)
Which of the halogens exist as a reddish-brown liquid at room temperature?
2. Question
1 point(s)
Which of the following is chemically most reactive among halogens?
3. Question
1 point(s)
Which of the following statements is TRUE about the Group 17 elements?
4. Question
1 point(s)
Which of the following statement is TRUE about group 17 elements.
5. Question
1 point(s)
Which of the following is correct about the characteristic of elements in group 17 in the periodic table as going down the group?
6. Question
1 point(s)
Both chlorine and bromine are elements in group 17 in the periodic table. The boiling point of chlorine is lower than bromine. Which statement below explains this phenomena?
7. Question
1 point(s)
Element X is located in the same group as bromine in the periodic table of elements. Which of the following is true about element X?
8. Question
1 point(s)
Element P dissolve in water to produce an acidic solution. The solution can turn litmus paper colourless. Which of the following is most likely to be P?
9. Question
1 point(s)
Which of the following is the product formed when bromine reacts with sodium hydroxide?
10. Question
1 point(s)
Which of the following is the product formed when chlorine gas dissolves in water?
11. Question
1 point(s)
Which of the following are the uses of chlorine?